 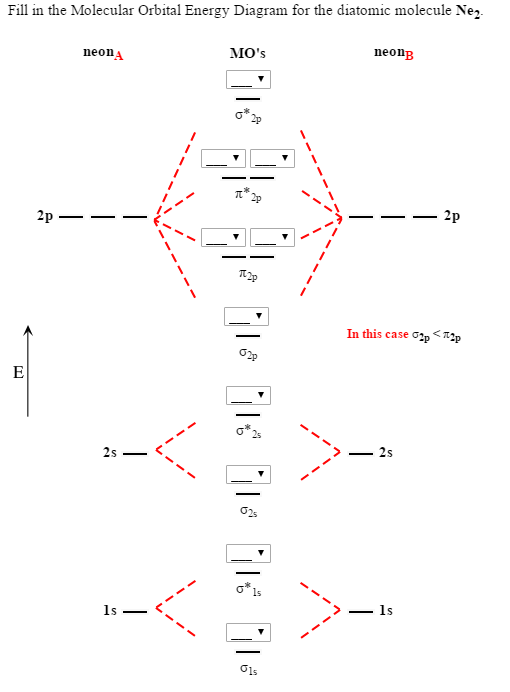
The molecular orbital corresponding to the sum of the two H 1 s orbitals is called a σ 1 s combination (pronounced “sigma one ess”) (part (a) and part (b) in Figure 9.18 "Molecular Orbitals for the H"). A molecule must have as many molecular orbitals as there are atomic orbitals.Īdding two atomic orbitals corresponds to constructive interference between two waves, thus reinforcing their intensity the internuclear electron probability density is increased. The molecular orbitals created from Equation 9.2 are called linear combinations of atomic orbitals (LCAOs) Molecular orbitals created from the sum and the difference of two wave functions (atomic orbitals). Consequently, the molecular orbital approach, called molecular orbital theory A delocalized bonding model in which molecular orbitals are created from the linear combination of atomic orbitals (LCAOs)., is a delocalized approach to bonding.Įquation 9.2 MO ( 1 ) = AO ( atom A ) + AO ( atom B ) MO ( 2 ) = AO ( atom A ) − AO ( atom B ) As the name suggests, molecular orbitals are not localized on a single atom but extend over the entire molecule. Just as the positions and energies of electrons in atoms can be described in terms of atomic orbitals (AOs), the positions and energies of electrons in molecules can be described in terms of molecular orbitals (MOs) A particular spatial distribution of electrons in a molecule that is associated with a particular orbital energy.-a spatial distribution of electrons in a molecule that is associated with a particular orbital energy. In Chapter 6 "The Structure of Atoms", we described the electrons in isolated atoms as having certain spatial distributions, called orbitals, each with a particular orbital energy. Just as with the valence bond theory, the approach we are about to discuss is based on a quantum mechanical model. 
Such limitations led to the development of a new approach to bonding in which electrons are not viewed as being localized between the nuclei of bonded atoms but are instead delocalized throughout the entire molecule. (For more information on semiconductors, see Chapter 12 "Solids", Section 12.6 "Bonding in Metals and Semiconductors".) These approaches also cannot describe the nature of resonance. None of the approaches we have described so far can adequately explain why some compounds are colored and others are not, why some substances with unpaired electrons are stable, and why others are effective semiconductors. To use molecular orbital theory to predict bond order. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
